How can we
help you?
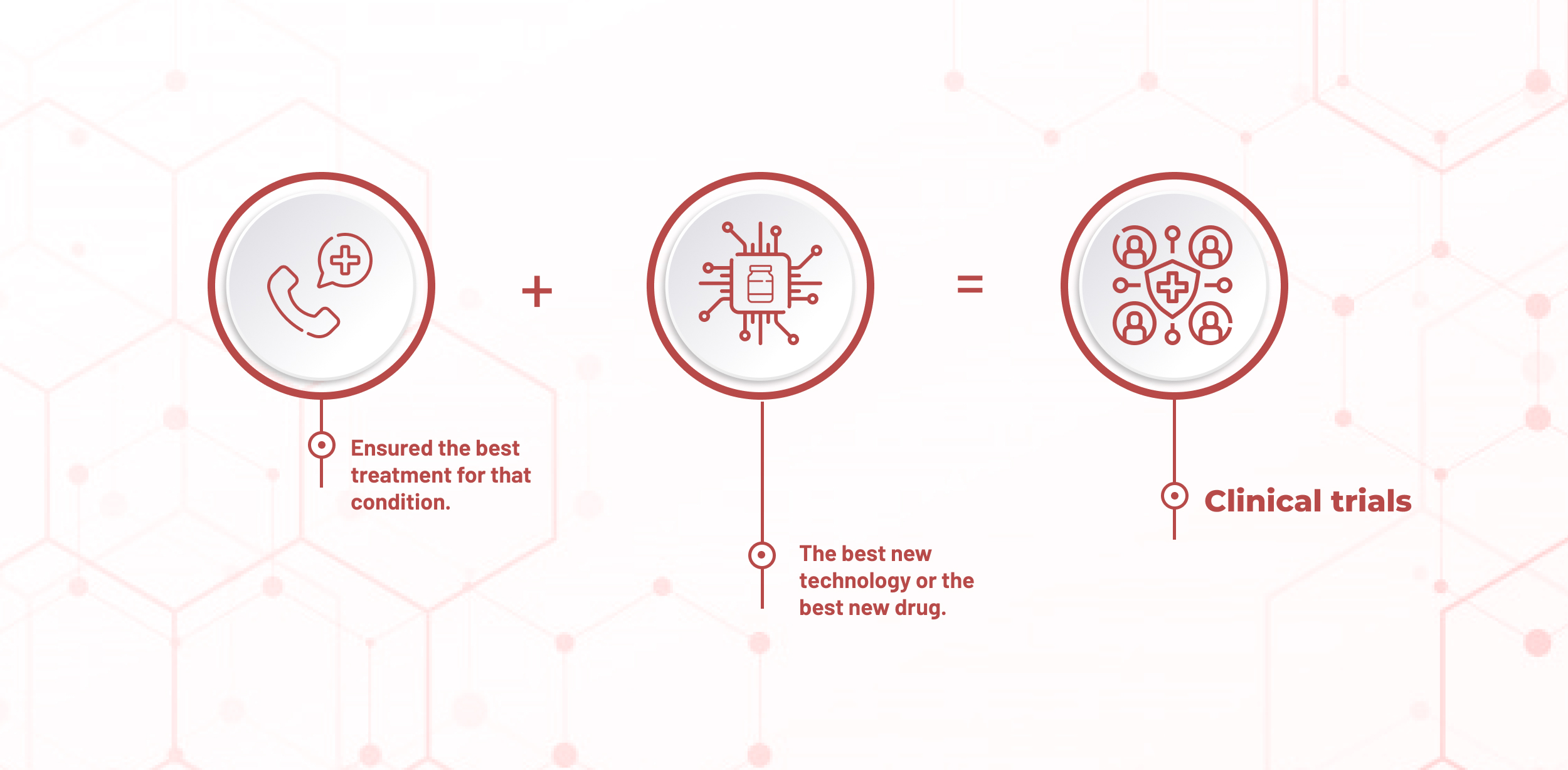
With us you can ask for a second opinion - we recommend, within your pathology, the best specialist!
We help you search for clinical trials and evaluate your inclusion/referral. We will accompany you during the evaluation and inclusion phase.






New Breathe Services


newbreathe
Requesting a second opinion, with the best specialist in the area, is now more practical and faster with our support.


newbreathe +
Finding out if clinical trials are available will be a simple process with the help of our team.


newbreathe global
Finding out if there are any clinical trials available for your condition, we’ll walk you through the whole process.
A Team of Professionals
A Team of Professionals



Pedro Nogueira da Silva



Francisco Cruz



Manuel Teixeira Gomes



Tiago Sotto Maior


João Massano



André Soares
Patient testimonials



After a thoughtless act I developed caustic esophagitis - as a consequence, I could not swallow anything at all, and there was the need for a feeding tube placed directly into my stomach.
My hospital and the field specialists that I contacted considered that it was an inoperable case. I contacted New Breathe who referred me to an esophageal surgery specialist who agreed to review my case and operate on me. The surgery was extremely complicated, I spent one month in intensive care, but progressively I have been recovering. Today I can already eat normally with my mouth, despite some dietary restrictions.
Anonymous



My father, at the age of 70, developed a rare and extremely aggressive leukemia. He was initially seen at the IPO and started chemotherapy, which went very well. For 7 months there was no evidence of disease. After this period, a skin lesion appeared and a biopsy confirmed a recurrence. Local radiotherapy was performed, but in a very short time there were new manifestations. Given the aggressiveness and low response to treatments, he was not offered any therapeutic alternative.
In the research that me and my brothers did together, we contacted New Breathe to evaluate possible alternatives. Within a short period of time they proposed the possibility of inclusion in a phase II clinical trial taking place in the United States. After we were accepted, they contacted the sponsor of the clinical trial and verified that my father met the criteria to eventually be included. They took care of all the paperwork, travel and lodging. My father was able to undergo an experimental treatment that allowed him a few more long months of virtually asymptomatic life.
Pedro
We have some
Good News


0-800-777-2331
24/7 EMERGENCY NUMBER
Call us now if you are in a medical emergency need, we will reply swiftly and provide you with a medical aid.